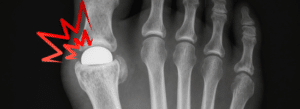
If you received a Cartiva Synthetic Cartilage Implant in your big toe and are now experiencing pain, implant failure, or need for revision surgery, you may be eligible to file a Cartiva toe implant lawsuit.
Reports, studies, and federal litigation suggest the Cartiva implant may have higher failure rates than originally disclosed, leading to serious complications for patients.
The Cartiva Synthetic Cartilage Implant (SCI) is a polyvinyl alcohol (PVA) hydrogel device designed to treat big toe arthritis (hallux rigidus) by preserving joint motion and avoiding fusion surgery.
It was marketed as:
However, real-world outcomes have raised significant concerns about safety, durability, and long-term effectiveness.
In October 2024, the U.S. Food and Drug Administration issued a Class II recall for Cartiva implants.
The recall cited higher-than-expected complication rates, including:
The recall applied to devices distributed between July 2016 and October 2024.
While early studies suggested relatively strong outcomes, post-market data tells a different story.
Reported issues include:
In many cases, patients ultimately required the fusion surgery the implant was meant to avoid.
The most common failure mechanism is subsidence, where the implant sinks into surrounding bone.
Other contributing factors may include:
These failures can lead to chronic pain, reduced mobility, and additional surgeries.
Patients frequently report:
If you are experiencing these symptoms after a Cartiva procedure, your case may align with known failure patterns.
If you or a loved one has been affected by the Cartiva Toe Implant, don’t wait to seek legal help.
Contact Salenger, Sack, Kimmel & Bavaro today for a FREE consultation.
Call (800) 675-8556 or contact us online to get started.
Cartiva litigation is expanding nationwide.
MDLs often signal that litigation has reached a stage where global settlements or bellwether trials may follow.
Cartiva lawsuits generally claim that manufacturers:
Claims typically include:
You may be eligible if:
Each case depends on medical records, but many affected patients meet these criteria.
Compensation may include:
Not every Cartiva implant fails. Some patients report improved mobility and pain relief, particularly in the short term.
However, the legal focus centers on whether:
This distinction is critical in product liability litigation and will likely shape how cases are resolved.
The Cartiva implant is a synthetic cartilage device made from polyvinyl alcohol hydrogel, designed to treat arthritis in the big toe while preserving joint motion.
The FDA issued a Class II recall due to higher-than-expected complication rates, including implant failure, migration, and chronic pain.
Common symptoms include persistent pain, stiffness, limited mobility, swelling, and the need for revision surgery.
Yes. Lawsuits are being filed nationwide, and a federal MDL was established in 2026 to coordinate cases.
Settlement amounts vary based on injury severity, medical costs, and long-term impact. There is no fixed average at this stage.
If you are dealing with complications from a Cartiva implant, a legal evaluation can help determine whether you have a viable claim.
Consultations are typically:
Because statutes of limitations apply, timely review of your case is important.
Contact Salenger, Sack, Kimmel & Bavaro today for a FREE consultation.
Call (800) 675-8556 or contact us online to get started.